Updated April 6, 2026, with final information; outbreak is over. Rosabella brand moringa powder capsules were the source of an “extensively drug-resistant” Salmonella outbreak that has ended after causing 10 illnesses and three hospitalizations, according to state and federal health officials. This outbreak is not associated with another ongoing Salmonella outbreak involving moringa leaf powder.
Do you need a Salmonella lawyer?
Which Products Pose a Risk?
In the US, Ambrosia Brands LLC distributes Rosabella moringa capsules. The U.S. Food and Drug Administration (FDA) has recommended that the firm issue a recall, but that has not yet happened.
Rosabella brand morinaga powder capsules are sold in white and green plastic jars. The affected products have expiration dates ranging from 3/2027 to 11/2027 and lot codes ranging from 5020591 to 5100048. To see a complete list, click here.

Rosabella Moringa Powder Drug-Resistant Salmonella Outbreak
Health officials confirmed 10 illnesses in eight states: AZ, FL, IA, IL, IN, TN, VA, and WA, but many more people were likely sickened.
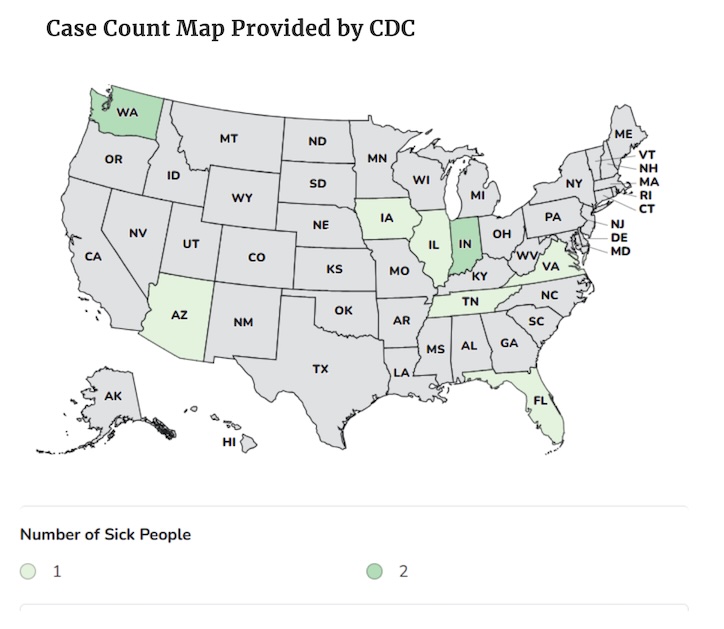
DNA fingerprinting tests on the Salmonella Newport cultured from all patients show they match. This means patients were exposed to the same source of contamination. Health officials interviewed eight of the patients; seven reported taking these capsules before they became ill. The FDA and state health departments are working to determine the source of the contamination.
Because all seven cultures of this Salmonella Newport outbreak strain show predicted resistance to 15 common antibiotics, it is considered extensively drug resistant (XDR). This means patients requiring treatment may need a different medicine.
Antibiotic-Resistant Salmonella Newport
This is the first time a U.S. outbreak has been linked to this “extensively drug-resistant strain of Salmonella Newport with an NDM-1 gene,” according to the Centers for Disease Control and Prevention (CDC).
XDR Salmonella shows resistance to:
- All first-line antibiotics
- Common alternative antibiotics
May also be resistant to:
- Multiple β-lactam antibiotics
The CDC advises doctors to consult an infectious disease specialist for treatment of patients with XDR Salmonella infections and monitor them for treatment failure.
Salmonella Symptoms
Symptoms of a Salmonella infection usually develop within six to 72 hours and include:
- Nausea
- Vomiting
- Abdominal cramps
- Diarrhea that is sometimes bloody
- Fever
Additional Information About Salmonella
- Salmonella Outbreak Lawsuit FAQ
- Drug-Resistant Salmonella in Raw Turkey Sickens 90
- Salmonella Lawsuit: 5 Reasons to Sue for Compensation
- Member’s Mark Super Greens Salmonella Outbreak [Update]
- Live it Up Super Greens Supplement Salmonella Outbreak
- Salmonella Symptoms
- Salmonella Typhimurium
Other Moringa Powder Salmonella Outbreaks
An ongoing Salmonella outbreak linked to the Live it Up Super Greens supplement, which contains moringa powder, has affected 65 people in 28 states. At least 14 people have been hospitalized. Thar outbreak has triggered a recall for all Live it Up supplements made since January 2025.
In December 2025, a Salmonella outbreak linked to Member’s Mark Super Greens sold at Sam’s Club stores ended after sickening 11 people in seven states. Three people were hospitalized.
Experienced Salmonella Lawyers
If you developed a Salmonella infection after consuming moringa capsules and would like a free consultation with an experienced Salmonella lawyer, please contact the Pritzker Hageman Salmonella Legal Team. Our attorneys have represented clients in every major Salmonella outbreak in the U.S. Call us at 1-888-377-8900, text 612-261-0856, or complete the form below. There is no obligation, and we don’t get paid unless we win.