At least 15 cancer patients contracted Exophiala dermatitidis fungal infections after receiving port implants and IV medications from one New York City oncology clinic, according to an article in this week’s Morbidity and Mortality Weekly Report, a CDC publication.
From January 1 to May 31, 2016, 38 patients were seen at the implicated oncology clinic (clinic A) and received IV medication. Of theses 38 patients, 29 patients were involved in the investigation. Of these 29, 15 were determined to have Exophiala dermatitidis infections. Two of the these patients died; one 10 days after testing positive, the other 12 weeks after testing positive. According to the CDC, it is unclear if the deaths were related to the infection.
The common denominator for the 15 patients who tested positive for Exophiala dermatitidis was exposure to “compounded” IV flush solution administered through a central venous catheter (CVC) (implanted port or peripherally inserted central catheter). The compounded IV flush solution contained saline, heparin, vancomycin, and ceftazidime, and it was compounded (mixed together) and administered at clinic A.
On May 24, 2016, the New York City Department of Health and Mental Hygiene notified the CDC of two cases of Exophiala dermatitidis bloodstream infections among cancer patients who had received care from a single physician at an outpatient oncology facility (clinic A). The CDC then conducted the investigation that led to finding 13 additional patients with Exophiala dermatitidis infections. Health officials looked at medical records and microbiology records for all patients who had received an IV medication at clinic A from January 1 to May 31, 2016.
What is Exophiala Dermatitidis?
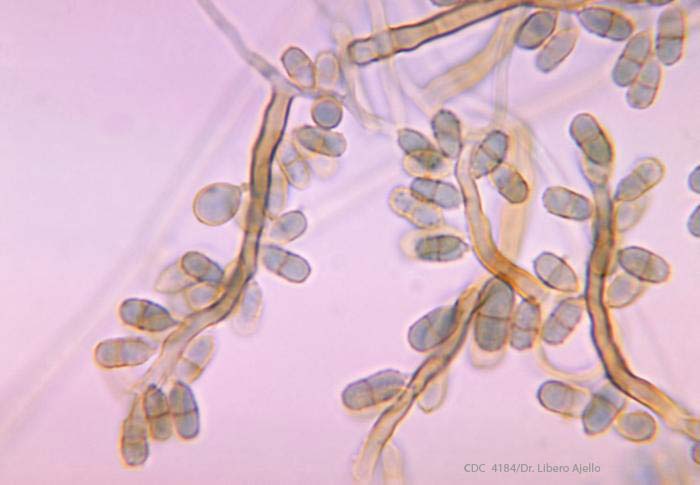
Exophiala dermatitidis is a fungus categorized as black yeast. Human infection with Exophiala dermatitidis is rare, particularly in the United States. The incubation period (time between transmission and illness) can be months. The mortality rate (number of people infected who die) is about 40%. Complications can include the following:
- skin lesions and abscesses; and
- infection of the eye, heart, lungs or brain (meningitis or abscess);
- sacroilitis (infection of one or both sacroiliac joints, located where the pelvis and lower spine meet);
- sepsis.
Patients with possible clinic-associated Exophiala infections should be reported to their state health department and to CDC.
Implicated Oncology Clinic Not Compliant with Infection Prevention Standards
Federal health officials conducted an assessment of clinic A. This assessment found:
- failures to meet CDC infection control standards for outpatient oncology settings
- failures to meet standards for sterile medication compounding and handling of hazardous drugs.
“Investigators learned that IV flush bags containing saline, heparin, vancomycin, and ceftazidime had been compounded under substandard conditions, stored in a refrigerator, and accessed daily for multiple patients over approximately 4–8 weeks until the solution was depleted” (Vasquez).
The outbreak investigation and assessment prompted the New York City Department of Health to issue an order on May 31, 2016, which required the medical provider to stop treating patients at clinic A until that facility became compliant with medication preparation and infection prevention standards.
Exophiala Dermatitidis Lawsuit
Our legal team has helped people who contracted severe, life-threatening, fungal infections from compounded medication.
“Patients and their families trust medical professionals to use safe medications,” said attorney Fred Pritzker. “When this does not happen, our job is to find out exactly how a medication got contaminated, if any testing was done to detect this, and why it was administered to patients. Patient safety should always come before profits and expediency.”
A lawsuit for compensation in cases involving fungal infections from tainted compounded medication can help patients get answers and justice.
Sources
Vasquez AM, Lake J, Ngai S, et al. “Notes from the Field: Fungal Bloodstream Infections Associated with a Compounded Intravenous Medication at an Outpatient Oncology Clinic — New York City, 2016.” MMWR Morb Mortal Wkly Rep 2016;65:1274–1275.
Centers for Disease Control and Prevention (CDC. “Exophiala infection from contaminated injectable steroids prepared by a compounding pharmacy–United States, July-November 2002.” MMWR. Morbidity and mortality weekly report 51.49 (2002): 1109.
Kenney, Richard T., et al. “Successful treatment of systemic Exophiala dermatitidis infection in a patient with chronic granulomatous disease.” Clinical infectious diseases 14.1 (1992): 235-242.